Chiropractor, Dr. Alexander Jimenez looks at the detailed anatomy and function of psoas major, and the role it plays in lumbo-pelvic stability.
The purpose of this two-part article therefore is to dissect the literature on the PM both in terms of its anatomy and its function, and outline the role it plays in lumbar spine stability and presenting pain and pathology in and around the lumbopelvic area.
Anatomy & Biomechanics
Proximal anatomy
The PM is a long unipennate muscle that has one fascial attachment (via the diaphragm) and two fibrous attachment onto the spine (see Figure 1)(2). In terms of its upper fascial attachment onto the lumbar spine and the diaphragm, the PM attaches to the diaphragm via the medial arcuate ligament. This is a tendinous arch in the fascia that covers the PM, with its fibres converging to a tendon that ascends to the diaphragm(2).
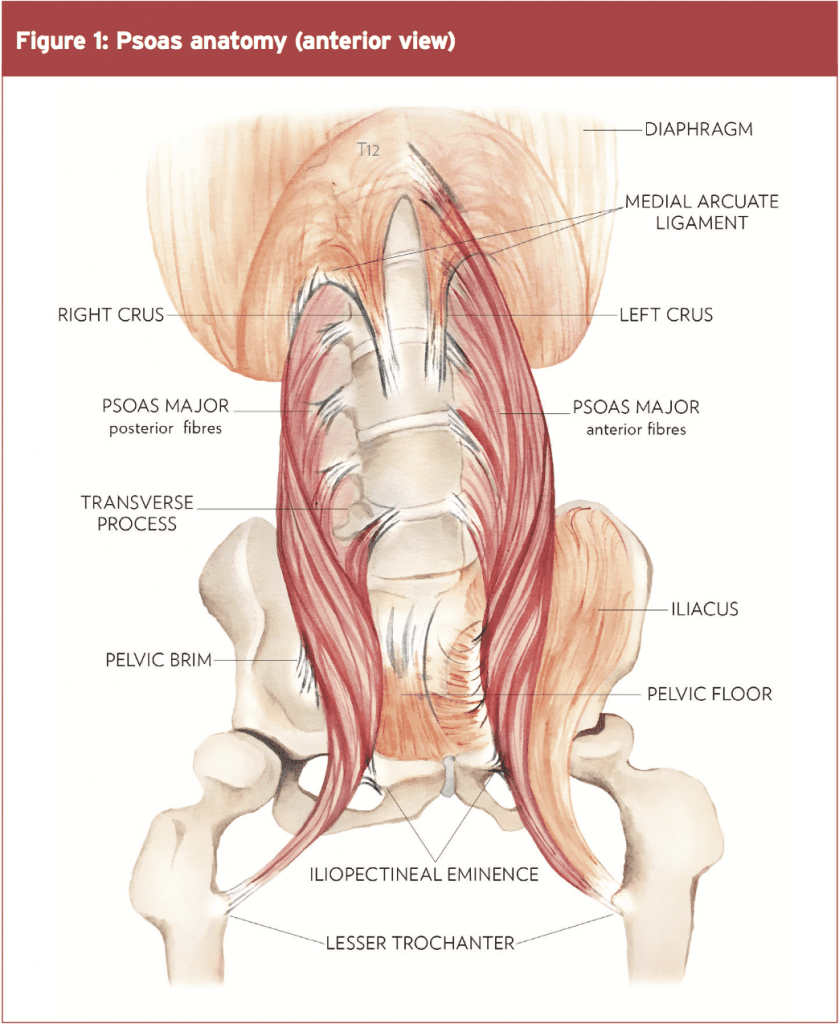
The diaphragm itself attaches to the spine via the right and left crus. They attach to the anterolateral component of the upper three lumbar vertebral bodies. The crus and their fascia overlap the PM and appear to be continuous with this muscle until they come more anterior and blend with the anterior longitudinal ligament of the spine(3). This PM-to-diaphragm attachment may have implications for lumbar spine stability as discussed later in the text.
The more traditional understanding of the proximal fibrous attachments is that the attachments of the PM are arranged as fascicles. They attach anteriorly to the L1 – L5 transverse processes (these are the posterior fascicles) and anteriorly from the T12 – L1 disc to the L4 – L5 disc (these are the anterior fascicles). The L5/S1 disc has no attachment(4).
The fibre length ranges between 3-5 cm within the posterior fascicles, and 3-8 cm in the anterior fascicles. The fascicles rotate medially so the posterior fascicles become anterior and the anterior become posterior as they descend down and laterally towards the pelvic rim(2,5). These short fascicle lengths mean that the fibre length does not extend the full length of the muscle.
In a more recent study, Arbanas et al (2009) explored the differences in the fibre type composition of the PM muscle between its cranial and caudal portion(20). They found that the fibre type composition of PM changed from the cranial towards the caudal part of its origin. The percentage of type I fibres had a decreasing trend (less type 1 fibres around the distal part of the muscle), whereas the percentage of type II fibres had an increasing trend (more type 2 fibres distally).
Based on these findings the authors suggested that the distal part of the PM could have a dynamic role whereas the proximal portion may have more of a postural role. This fits with the descriptions of the posterior PM having more of a stability role and the anterior fibres having more of a mobility role at the hip(2).

There is growing evidence showing how these muscles coordinate their activity to stabilise the spine. It has been shown that the transversus abdominus co-contracts with the diaphragm(43), the pelvic floor(44,45) and the deep fibres of lumbar multifidus(11). According to this model, the PM is ideally located to assist in a stabilising role.
An extremely useful analogy to describe trunk stability is the ‘cylinder effect’ created by all of the associated muscles (see Figure 2). The top of the cylinder is the diaphragm, the bottom is the pelvic floor and the middle is the transversus abdominis muscle, with the thoracolumbar fascia. PM is ideally suited to act as a link between the top and bottom of the cylinder through the medial arcuate ligament to the diaphragm and the PM fascia to the pelvic floor.

This kind of mechanical stability is used in engineering. The placement of a rod in a cylinder will resist displacement in all directions(2,46). In the PM analogy, this can be thought of conceptually as a supporting rod in the middle of the cylinder. Early biomechanical literature suggested that the PM might aid in the stabilisation of the lumbar spine through its large potential to generate compressive forces, which would result in increased spinal stiffness(47).
McGill 2002 conceptualizes lumbar spine stability as a fishing rod placed upright and vertical with tensioned guy wires attached at different levels along its length and those guy wires being attached
to the ground in a circular pattern(42). Here the rod represents the lumbar vertebrae and the guy wires are the various muscles attaching to the lumbar spine. Reducing the tension on one of the muscles (wires) will allow the spinal segment (rod) to buckle and allow spinal injury to occur.
Juker et al (48) showed that the PM counteracts the action of iliacus during hip flexion(48). They believe that the iliacus would torque the pelvis into anterior pelvic tilt and that the PM works against these forces, adding to the stiffness within the pelvis and the lumbar spine. An activated and stiffened PM will contribute some shear stiffness to the lumbar motion segment (49,50). This last point poses an interesting question. Does the PM contract to create hip flexion or does it contract to counterbalance the anterior tilt force imposed by contraction of the other hip flexors?
References
1. Manuelle Therapie. 2007. 11:177-187
2. Proceedings of: 11th Annual National
Orthopaedic Symposium. “The Tragic Hip:
Trouble in the Lower Quadrant”. Nov 6-7 1999,
Halifax, Canada.
3. Arch Phys Med Rehab. 1994; 75:703–708
4. Clin Biomech, 1992. 7:109–119
5. Proceedings of: 4th Interdisciplinary World
Congress on Low Back Pain. November 9-11
2001, Montreal, Canada.
6. Williams PL (1995) Gray’s Anatomy: The
Anatomical Basis of Medicine and Surgery.
Edinburgh: Churchill Livingstone.
7. Rickenbacker J, Landholt AM, Theiler K
(1985) Applied Anatomy of the Back.
Heidelberg: Springer.
8. Bogduk N and Twomey LT (1997) Clinical
Anatomy of the Lumbar Spine and Sacrum.
London: Churchill Livingstone.
9. Orthopaedic Division Review. 2002. Jan /
Feb: 7-16
10. Williams PL, Warwick R, Dyson M and
Bannister 1989 Gray’s Anatomy 37th ed.
Churchill Livingstone, Edinburgh
11. Spine 2002. 27(2):E29-E36
12. Histochemistry. 1992. 97, 77–81
13. Acta Radiol. 1993. 24, 16–19
14. Z Morphol Anthropol. 2002. 83, 305–314
15. Clin Physiol. 1989. 9, 333–343
16. Acta Physiol Scand. 1983. 117, 115–122
17. Anat Rec. 1982. 203, 451–459
18. J Neurol Sc. 1984. 63, 85–100
19. J Anat. 1999. 194, 355–362
20. J. Anat. 2009. 215, pp636–641
21. Anatomical Record. 1968. 160, 310–311
22. Scand J Med Sci Sports. 1995. 5:10-16
23. Walther DS (1981) Pelvis and thigh muscles.
In Applied Kinesiology, Vol. I, pp. 302–305
24. Clin Anat. 1999. 12, 264–265
25. Scand J Med Sci Sports. 1995. 5:10–16
26. Physical Therapy. 1986. 66(3):351-361
27. Orthopaedic Clinics of North America. 1977.
8(1):193-199
28. Journal of Biomechanics. 1995. 28:339-
345
29. Journal of Orthopedic Medicine. 1991.
13(2):34-40
30. Spine: State of the Art Reviews. 1995.
9(2):419-432
31. Proceedings of: 1st International
Conference on Movement Dysfunction. Sept
21-23, 2001. Edinburgh, Scotland.
32. Journal of Orthopaedic Science. 2002. 7:
199 – 20
33. Romanes GJ (1981) Cunningham’s Textbook
of Anatomy. Toronto: Oxford University Press.
34. Kapandji IA (1974) The Physiology of the
Joints, vol. 3. New York: Churchill Livingstone.
35. Research Quarterly for Exercise and Sport.
1980. 51, 334–348
36. European Journal of Applied Physiology.
1985. 54, 524–532
37. Acta Orthop Scand. 1966. 37:177–190
38. Acta Orthopaedica Scandinavica. 1968.
39:47-57
39. Nordin M and Frankel V (1989) Basic
Biomechanics of the Musculosceletal System.
Malvern: Lea & Febiger.
40. Crisco JJ, Panjabi MM (1990) Postural
biomechanical stability and gross muscular
architecture in the spine. In Multiple Muscle
Systems: Biomechanics and Movement
Organization (ed. Winters J, Woo S). New York:
Springer.
41. Journal of Orthopedic Research. 1991.
19:228-236
42. McGill S 2002 Low Back Disorders.
Evidence-Based Prevention and Rehabilitation.
Human Kinetics, Champaign, IL
43. J Appl Physiol. 1997; 83(3):753–760
44. Arch Phys Med Rehab. 2001; 82(8):1081–
1088
45. Neurology and Urodynamics. 2001. 20:31-
42
46. Richardson C, Jull G, Hides J, Hodges P
1999 Therapeutic Exercise for Spinal
Stabilisation: Scientific basis and practical
techniques. Churchill Livingstone, Edinburgh
47. Spine. 2004; 29(22):E515–E519
48. Medicine & Science In Sports & Exercise.
1998. 30(2):301-310
49. Spine. 1998; 23(18):1937–1945
50. Spine. 1995; 20(2):192–198
The posterior fascicles are smaller, less vertical and are located very close to the axis of rotation. As the fibres rotate medially and end up in an anterior position, these fibres now come into contact with the psoas fascia. The anterior fascicles are larger, more vertical and more suited for acting on the hip as a hip flexor along with iliacus (see function below).
The psoas minor muscle (PMI) attaches on the anterior surface of the 12th thoracic and the first lumbar vertebrae(6), and may be absent in 50% of subjects(7). Due to its absence in a large percentage of the population, it will not be discussed in this article.
The PM does not simply pass over the pelvic brim like a pulley. As it crosses the iliopectineal eminence, the fascia of the posterior fascicles attach firmly to the pelvic brim. This may constitute an innominate ligament that binds the PM to the pelvis(9). As the PM descends downwards from the lower lumbar spine levels, its infero-medial fascia becomes thick at its inferior portion and is continuous with the pelvic floor fascia(2,10). This forms a link with the conjoint tendon, transverse abdominus, and the internal oblique muscle. Again, this PM to pelvic floor fascia arrangement with the pelvic floor and transversus abdominus may have implications for its role as a lumbo-pelvic stabiliser.
Therefore, it is possible that the distal PM has many attachments. These include:
The psoas minor muscle (PMI) attaches on the anterior surface of the 12th thoracic and the first lumbar vertebrae(6), and may be absent in 50% of subjects(7). Due to its absence in a large percentage of the population, it will not be discussed in this article.
Distal Anatomy
The exact insertion of the PM has recently received some attention. The traditional model of the PM has been that the tendon coursed infero-laterally, and joined the iliacus tendon to form the common iliopsoas tendon, and attached onto the lesser trochanter of the femur(6,8). However, more recent research suggests that the distal anatomy may be a lot more complex than originally perceived.The PM does not simply pass over the pelvic brim like a pulley. As it crosses the iliopectineal eminence, the fascia of the posterior fascicles attach firmly to the pelvic brim. This may constitute an innominate ligament that binds the PM to the pelvis(9). As the PM descends downwards from the lower lumbar spine levels, its infero-medial fascia becomes thick at its inferior portion and is continuous with the pelvic floor fascia(2,10). This forms a link with the conjoint tendon, transverse abdominus, and the internal oblique muscle. Again, this PM to pelvic floor fascia arrangement with the pelvic floor and transversus abdominus may have implications for its role as a lumbo-pelvic stabiliser.
Therefore, it is possible that the distal PM has many attachments. These include:
- The iliopectineal eminence (innominate ligament).
- The infero-medial fascia, which blends with the pelvic floor fascia via the PM’s inferomedial fascia (this then links with the transversus abdominus fascia).
- Anterior fibres, which may be the fibres that continue on and join the iliacus muscle.
Nerve Supply
The nerve supply of PM is segmental. The anterior and posterior fascicles of PM have a separate nerve supply. The posterior fascicles are supplied by the ventral rami of spinal nerves T12 through L4. The anterior fascicles are supplied by branches of the femoral nerve from L2, 3 and 4(2). This segmental innervation may be analogous with the lumbar multifidus muscles, which have been shown to have separate functional roles in spinal stability(11).Fibre Types
There are disagreements in the literature about whether the PM muscle is mostly composed of slow-twitch or fast-twitch fibres. It was originally believed that the PM muscle had a heterogeneous distribution of fibre types across the muscle(12-14). However, further cadaver studies that enable an analysis of the entire PM muscle have shown that the size of the muscle fibres and the distribution of the muscle fibre types vary according to location(15,16). Differences can be found with regard to the superficial or deeper parts, as well as the proximal or distal parts of the muscle(17-19).In a more recent study, Arbanas et al (2009) explored the differences in the fibre type composition of the PM muscle between its cranial and caudal portion(20). They found that the fibre type composition of PM changed from the cranial towards the caudal part of its origin. The percentage of type I fibres had a decreasing trend (less type 1 fibres around the distal part of the muscle), whereas the percentage of type II fibres had an increasing trend (more type 2 fibres distally).
Based on these findings the authors suggested that the distal part of the PM could have a dynamic role whereas the proximal portion may have more of a postural role. This fits with the descriptions of the posterior PM having more of a stability role and the anterior fibres having more of a mobility role at the hip(2).

PM As A Lumbo-Pelvic Stabiliser
Due to the unique anatomy of the PM as described in box 1, it is possible that the PM has both a hip flexion role (anterior fascicles) and a very important spine stabilisation role (posterior fascicles)(2). PM has intimate anatomical attachments to the diaphragm and the pelvic floor. This unique anatomical location allows the PM to act as a link between these structures and may help in maintaining the stability of the ‘lumbar cylinder’ mechanism.There is growing evidence showing how these muscles coordinate their activity to stabilise the spine. It has been shown that the transversus abdominus co-contracts with the diaphragm(43), the pelvic floor(44,45) and the deep fibres of lumbar multifidus(11). According to this model, the PM is ideally located to assist in a stabilising role.
An extremely useful analogy to describe trunk stability is the ‘cylinder effect’ created by all of the associated muscles (see Figure 2). The top of the cylinder is the diaphragm, the bottom is the pelvic floor and the middle is the transversus abdominis muscle, with the thoracolumbar fascia. PM is ideally suited to act as a link between the top and bottom of the cylinder through the medial arcuate ligament to the diaphragm and the PM fascia to the pelvic floor.

This kind of mechanical stability is used in engineering. The placement of a rod in a cylinder will resist displacement in all directions(2,46). In the PM analogy, this can be thought of conceptually as a supporting rod in the middle of the cylinder. Early biomechanical literature suggested that the PM might aid in the stabilisation of the lumbar spine through its large potential to generate compressive forces, which would result in increased spinal stiffness(47).
McGill 2002 conceptualizes lumbar spine stability as a fishing rod placed upright and vertical with tensioned guy wires attached at different levels along its length and those guy wires being attached
to the ground in a circular pattern(42). Here the rod represents the lumbar vertebrae and the guy wires are the various muscles attaching to the lumbar spine. Reducing the tension on one of the muscles (wires) will allow the spinal segment (rod) to buckle and allow spinal injury to occur.
Juker et al (48) showed that the PM counteracts the action of iliacus during hip flexion(48). They believe that the iliacus would torque the pelvis into anterior pelvic tilt and that the PM works against these forces, adding to the stiffness within the pelvis and the lumbar spine. An activated and stiffened PM will contribute some shear stiffness to the lumbar motion segment (49,50). This last point poses an interesting question. Does the PM contract to create hip flexion or does it contract to counterbalance the anterior tilt force imposed by contraction of the other hip flexors?
In summary
The PM should be regarded as having a very unique and ingenious anatomical design. It allows the spine to remain stiff and stable via its compressive effect on the spine and due to its contribution with other ‘core’ muscles to maintain the internal ‘balloon’ effect in the abdominal wall. Furthermore, it may contribute to hip flexion, as well as maintaining pelvic stability during hip flexion to prevent the anterior tilt drag effect created by the other hip flexors. Clearly when the demands of the hip and pelvis are increased during high-level sport function, this mechanism will automatically augment stability in the lumbopelvic region along with a greater contribution from the more superficial abdominal wall muscles. In the next article, pain and dysfunction in the PM will be discussed, and retraining of the PM muscle in the light of its functional requirements will be explored.References
1. Manuelle Therapie. 2007. 11:177-187
2. Proceedings of: 11th Annual National
Orthopaedic Symposium. “The Tragic Hip:
Trouble in the Lower Quadrant”. Nov 6-7 1999,
Halifax, Canada.
3. Arch Phys Med Rehab. 1994; 75:703–708
4. Clin Biomech, 1992. 7:109–119
5. Proceedings of: 4th Interdisciplinary World
Congress on Low Back Pain. November 9-11
2001, Montreal, Canada.
6. Williams PL (1995) Gray’s Anatomy: The
Anatomical Basis of Medicine and Surgery.
Edinburgh: Churchill Livingstone.
7. Rickenbacker J, Landholt AM, Theiler K
(1985) Applied Anatomy of the Back.
Heidelberg: Springer.
8. Bogduk N and Twomey LT (1997) Clinical
Anatomy of the Lumbar Spine and Sacrum.
London: Churchill Livingstone.
9. Orthopaedic Division Review. 2002. Jan /
Feb: 7-16
10. Williams PL, Warwick R, Dyson M and
Bannister 1989 Gray’s Anatomy 37th ed.
Churchill Livingstone, Edinburgh
11. Spine 2002. 27(2):E29-E36
12. Histochemistry. 1992. 97, 77–81
13. Acta Radiol. 1993. 24, 16–19
14. Z Morphol Anthropol. 2002. 83, 305–314
15. Clin Physiol. 1989. 9, 333–343
16. Acta Physiol Scand. 1983. 117, 115–122
17. Anat Rec. 1982. 203, 451–459
18. J Neurol Sc. 1984. 63, 85–100
19. J Anat. 1999. 194, 355–362
20. J. Anat. 2009. 215, pp636–641
21. Anatomical Record. 1968. 160, 310–311
22. Scand J Med Sci Sports. 1995. 5:10-16
23. Walther DS (1981) Pelvis and thigh muscles.
In Applied Kinesiology, Vol. I, pp. 302–305
24. Clin Anat. 1999. 12, 264–265
25. Scand J Med Sci Sports. 1995. 5:10–16
26. Physical Therapy. 1986. 66(3):351-361
27. Orthopaedic Clinics of North America. 1977.
8(1):193-199
28. Journal of Biomechanics. 1995. 28:339-
345
29. Journal of Orthopedic Medicine. 1991.
13(2):34-40
30. Spine: State of the Art Reviews. 1995.
9(2):419-432
31. Proceedings of: 1st International
Conference on Movement Dysfunction. Sept
21-23, 2001. Edinburgh, Scotland.
32. Journal of Orthopaedic Science. 2002. 7:
199 – 20
33. Romanes GJ (1981) Cunningham’s Textbook
of Anatomy. Toronto: Oxford University Press.
34. Kapandji IA (1974) The Physiology of the
Joints, vol. 3. New York: Churchill Livingstone.
35. Research Quarterly for Exercise and Sport.
1980. 51, 334–348
36. European Journal of Applied Physiology.
1985. 54, 524–532
37. Acta Orthop Scand. 1966. 37:177–190
38. Acta Orthopaedica Scandinavica. 1968.
39:47-57
39. Nordin M and Frankel V (1989) Basic
Biomechanics of the Musculosceletal System.
Malvern: Lea & Febiger.
40. Crisco JJ, Panjabi MM (1990) Postural
biomechanical stability and gross muscular
architecture in the spine. In Multiple Muscle
Systems: Biomechanics and Movement
Organization (ed. Winters J, Woo S). New York:
Springer.
41. Journal of Orthopedic Research. 1991.
19:228-236
42. McGill S 2002 Low Back Disorders.
Evidence-Based Prevention and Rehabilitation.
Human Kinetics, Champaign, IL
43. J Appl Physiol. 1997; 83(3):753–760
44. Arch Phys Med Rehab. 2001; 82(8):1081–
1088
45. Neurology and Urodynamics. 2001. 20:31-
42
46. Richardson C, Jull G, Hides J, Hodges P
1999 Therapeutic Exercise for Spinal
Stabilisation: Scientific basis and practical
techniques. Churchill Livingstone, Edinburgh
47. Spine. 2004; 29(22):E515–E519
48. Medicine & Science In Sports & Exercise.
1998. 30(2):301-310
49. Spine. 1998; 23(18):1937–1945
50. Spine. 1995; 20(2):192–198




